Artificial Cornea Restores Perfect Vision in Untransplantable Eye
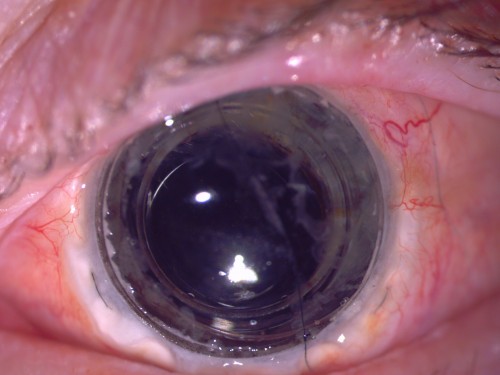 Israeli medtech pioneer CorNeat Vision has achieved a breakthrough in ophthalmic innovation. Their synthetic corneal implant, the CorNeat KPro, has successfully restored perfect 20/20 vision (6/6 or 6/6 metric) in a patient whose eye had been blinded by corneal scarring from shingles and was deemed unsuitable for traditional corneal transplantation. The patient has maintained stable, flawless vision for 11 months following the procedure, with the eye also exhibiting a remarkably natural, healthy appearance.
Israeli medtech pioneer CorNeat Vision has achieved a breakthrough in ophthalmic innovation. Their synthetic corneal implant, the CorNeat KPro, has successfully restored perfect 20/20 vision (6/6 or 6/6 metric) in a patient whose eye had been blinded by corneal scarring from shingles and was deemed unsuitable for traditional corneal transplantation. The patient has maintained stable, flawless vision for 11 months following the procedure, with the eye also exhibiting a remarkably natural, healthy appearance.
This achievement is particularly significant because the patient was never a candidate for a donor-dependent corneal transplant. CorNeat’s device circumvents this constraint by integrating directly into the eyewall beneath the conjunctiva, eliminating reliance on donor tissues. The fully synthetic design ensures long-term stability and visual performance without the risks of immune rejection.
Professor Irit Bahar, Head of Ophthalmology at Beilinson Hospital, heralded the outcome as a vivid testament to the transformative impact of technological breakthroughs in eye care, noting the profound implications for patients who previously had no restorative options.
"This is a landmark achievement," said Dr. Gilad Litvin, CorNeat Vision's Co-Founder and Chief Medical Officer. "Delivering sight and aesthetics to an eye once beyond help highlights the sheer impact of our artificial cornea. This breakthrough not only restores vision for patients with untreatable corneal conditions but also delivers hope to millions of blind patients living in regions where donor tissue is scarce."
CorNeat Vision’s CEO, Almog Aley-Raz, is set to present the CorNeat KPro and the interim findings of the RESEE clinical trial at the European Society of Cataract and Refractive Surgery (ESCRS) Innovation Day on 12 September in Copenhagen. The RESEE trial, which aims to secure both FDA 510(k) clearance and CE marking, is currently underway across clinical centres in Canada, France, India, Israel and the Netherlands, with plans to expand into the United States in 2026.
Supported by both the European Innovation Council and a U.S. Army Project Award, this initiative is rapidly gaining global traction, and the long shelf life and tissue-independent nature of the CorNeat KPro make it commercially appealing for distribution and adoption in diverse healthcare markets.