Breakthrough Retinal Implant Restores Central Vision in Advanced Macular Degeneration
 Researchers at the University Hospital Bonn have reported a world-first breakthrough in restoring central vision in patients with advanced age-related macular degeneration (AMD), using an innovative subretinal microchip implant. The findings, published in the New England Journal of Medicine (NEJM), demonstrate partial restoration of visual function in individuals previously affected by geographic atrophy — the severe late stage of AMD.
Researchers at the University Hospital Bonn have reported a world-first breakthrough in restoring central vision in patients with advanced age-related macular degeneration (AMD), using an innovative subretinal microchip implant. The findings, published in the New England Journal of Medicine (NEJM), demonstrate partial restoration of visual function in individuals previously affected by geographic atrophy — the severe late stage of AMD.
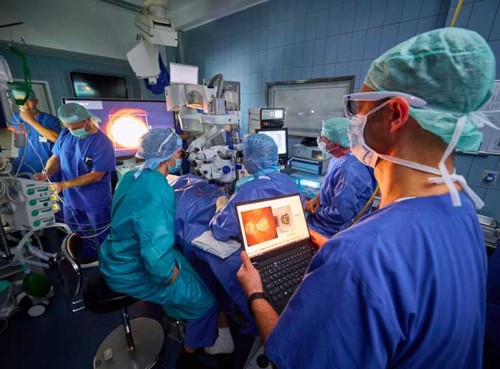
The international clinical trial involved 38 participants across 17 clinics in five countries. Each received the PRIMA implant, a 2 × 2 mm, 30-micrometre thin photovoltaic microchip surgically placed beneath the retina. The implant replaces the function of degenerated photoreceptor cells, converting projected infrared light into electrical signals that stimulate the remaining intact retinal cells. These signals are then processed by the brain’s visual cortex, enabling visual perception.
Participants use a specialised camera and infrared-projection glasses to transmit visual information to the implant. Remarkably, over 80% of participants experienced measurable improvements in visual acuity within 12 months, with more than 84% able to recognise letters, numbers or words after the procedure. Importantly, peripheral vision remained unaffected.
“This study marks a milestone in the treatment of geographic atrophy,” said Professor Frank Holz, lead author, retinal surgeon, and Director of the Department of Ophthalmology at University Hospital Bonn. “For the first time, we are seeing partial recovery of central vision in people with advanced AMD. This opens a completely new therapeutic perspective for patients who previously had no restorative options.”
Dr Mahi Muquit, retinal surgeon at London’s Moorfields Eye Hospital and co-author of the study, emphasised the significance of the findings: “While current therapies can slow disease progression, this approach represents a genuine paradigm shift — enabling partial restoration of sight where it was previously lost.”
An independent Data Safety Monitoring Board unanimously recommended the technology for approval on the European market, concluding that the clinical benefits outweigh potential risks. The implant was well tolerated overall, with most adverse events occurring within the first eight weeks post-surgery.
The PRIMA system, developed for patients with advanced dry AMD and central vision loss, is now undergoing regulatory review in Europe. Future iterations aim to further enhance image processing, comfort, and long-term stability.
The original research article, “Subretinal Photovoltaic Implant to Restore Vision in Geographic Atrophy Due to AMD,” by Frank G. Holz et al., is available in the New England Journal of Medicine (DOI: 10.1056/NEJMoa2501396).
(photo credit: Universitätsklinikum Bonn (UKB) / J.F. Saba)